RFID Mother-Infant Bracelets
Enhancing Safety and Connectivity in Maternal Care
REQUEST FOR QUOTATION
Description
RFID Mother-Infant Bracelets
RFID Technology Use – RFID bracelets use radio-frequency identification to securely link mother and newborn in healthcare settings.
Real-Time Tracking – These bracelets enable real-time tracking of mother and newborn to ensure safety and immediate identification.
Enhanced Communication – RFID systems improve communication among healthcare providers to reduce risks of mismatching and errors.
Key Technical Features
 Secure RFID Tags
Secure RFID Tags
The bracelets have secure RFID tags enabling reliable identification and data exchange in hospital settings.
Safe, Waterproof and Durable Materials
Made with waterproof and durable materials, the bracelets withstand harsh hospital environments.
Unique Identification Codes
Each bracelet includes unique identification codes for precise patient tracking and safety.
Wireless Connectivity
Wireless connectivity allows seamless data transmission and integration with hospital systems.
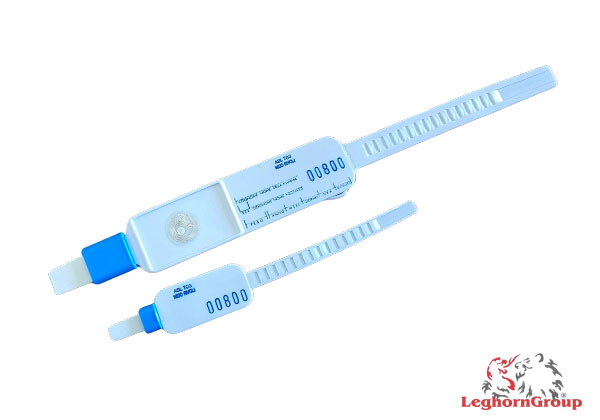
Security mother- infant bracelets
Bracelets with identical sequential numbering – unique RFID code – made of soft plastic with rounded edges, connected to each other by a thin cord that must be removed from the mother’s wrist at the moment of the baby’s birth.
This provides peace of mind, knowing that both healthcare professionals and the mother are able to immediately, electronically and visually verify that both bracelets are identically numbered. This numbering is sequential and in ascending order in the dispenser packs.
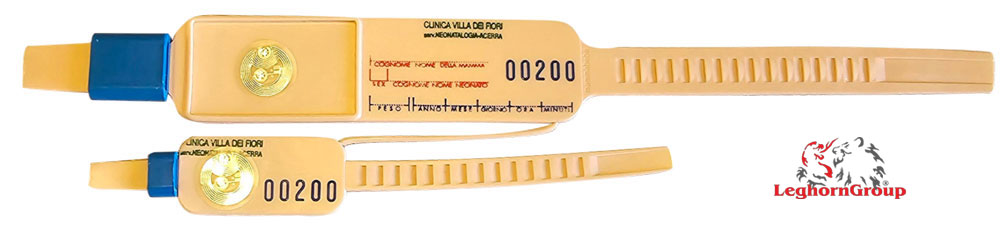
Security mother- infant bracelets
Safety closure with clips designed and manufactured from rigid plastic for removability in accordance with rigorous testing and inspection procedures.
The adaptability to the wrist is a unique feature of this identification system, as it allows the bracelet to be tightened after the baby’s physiological weight decreases. The rationally arranged spaces allow for data addition and support for computerization with a barcode or microchip.
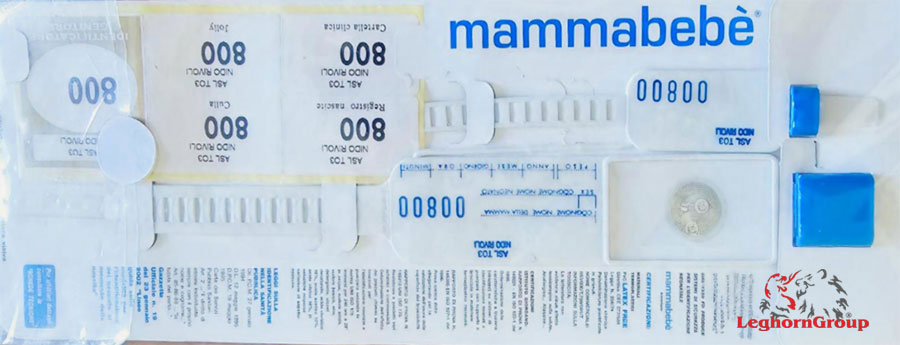
The kit
The Security Identifier Kit includes:
- Three identification bracelets, baby, mother and father or other person, with the same color impressed number and the same header hospital, to join the scientific and medical data.
These instruments allow to ensure certain relation between Registered Birth and Medical Records as indicated by the Ministry of Healthcare Guidelines.
SET COMPONENTS:
- 1 Pre-printed mother-baby numbered and personalized
- 5 Pre-numbered and personalized stickers (for birth register, crib, medical records, spare and parent identification)
- 1 Special permanent pen – can be used directly on the bracelet
Security mother- infant bracelets: how to use

- The two bracelets, originally joined (1), are separated and applied to the mother and newborn immediately after birth.
- Each mother and newborn is given a bracelet that displays, in addition to the hospital facility identification code (2), the same pre-printed sequential identification number (3), same as unique RFID code of the bracelet RFID inlay.
- The same sequential identification number (3) is reported on the birth register along with the data relating to the event, as well as on the medical records of the mother and newborn. This operation is facilitated by using pre-numbered stickers provided to the mother-to-be.
- Each birth center may, at its discretion, add other identifying elements in addition to the sequential number (data relating to the mother, the newborn’s date of birth, (4) etc.)
Functions and Operational Benefits
Automatic Identification – Bracelets enable quick and accurate automatic identification of patients to enhance safety and reduce errors.
Prevention of Infant Mix-Ups – The bracelets help prevent infant mix-ups by ensuring accurate matching between babies and mothers.
Emergency Alert System – Bracelets enable swift emergency alerts, allowing medical staff to respond promptly to urgent situations.
Improved Workflow Efficiency – These bracelets streamline nursing workflows, saving time and improving patient care quality.
Applicable Certifications and Compliance Standards
Medical Device StandardsISO 13485 sets quality management standards specifically for medical devices ensuring consistent safety and performance.
Regulatory ApprovalEU guidelines provide regulatory framework for safety and effectiveness of medical devices in the healthcare market.
Electromagnetic ComplianceCE certifications ensure electromagnetic compatibility of wireless medical devices like RFID bracelets for safe usage.
Product Quality Insurances
Codified and certified components, materials, and production processes.
Certifications:
- food-grade, latex-free, non-toxic, and hypoallergenic plastic materials (EU Directive 677/584, Law 256/74; test compliance EN-ISO 527-1 and UNI ISO 175).
- safety of the lettering against wear and liquids;
- certified tear resistance;
- customization of all variable characteristics.
Implementation in Hospital Environments
 Integration into Care Protocols
Integration into Care Protocols
Hospitals incorporate bracelets into maternal care protocols to improve patient safety and tracking accuracy.
Staff Training
Medical staff receive training to effectively use and manage the bracelets in clinical workflows.
System Connectivity
Bracelets are connected to hospital information systems to enhance monitoring and record keeping.